Sodium Hydroxide belongs to the category of inorganic compounds and is usually found in the form of a white solid at room temperature. This chemical compound consists of sodium Na + cations and hydroxide OH − anions. The Sodium Hydroxide chemical formula is written as NaOH. It is also commonly known as caustic soda or Iye and is widely used in manufacturing a variety of products such as paper, soap and detergents, pulp, explosives, liquid drain and oven cleaners, etc.
Sodium Hydroxide is one of the simplest hydroxides. Due to such characteristics, it is often used with neutral water and (acidic) HCl acid to determine the pH scale. We will learn more about Sodium Hydroxide, its properties, preparation, reaction, and uses below.
| IUPAC Name | Sodium Hydroxide / Sodium oxidanide |
| Chemical Formula | NaOH |
| Molar Mass | 39.997 g/mol |
| Density | 2.13 g/cm³ |
| Melting Point | 318.4 o C |
| Boiling Point | 1.388 °C |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 0 |
| Isotope Atom Count | 0 |
| Covalently-Bonded Unit Count | 2 |
Let us look at some of the properties of Sodium Hydroxide.
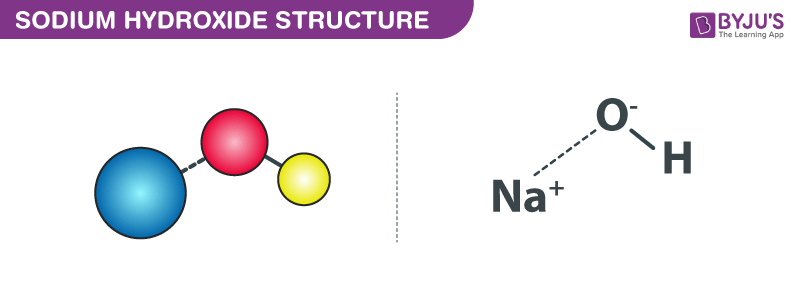
The structure of Sodium Hydroxide is represented by the following diagram:

Sodium hydroxide (NaOH) is one of the strongest bases available. It is one of the most important Alkali hydroxides used for laboratory and commercial purposes. Generally, over the years, sodium hydroxide has been manufactured on a large scale from readily obtainable raw material, and thus finds its use in numerous industrial processes. Predominantly, it is prepared by electrolysis of brine solution in a diaphragm or Mercury cell. However, there are also a few other important processes through which NaOH is produced along with other products (Cl2, Na2CO3, etc.,).
In this, the major product is chlorine gas but NaOH is produced as a side product. The overall equation of Leblanc process goes like this;
\(\beginHere, MnO2 acts as an oxidising agent, oxidising HCl to Cl2. In the olden days, preparation of HCl was difficult. It is prepared from NaCl by the action of a strong acid
Then, HCl produced is oxidised
The side product Na2SO4 is used for making other chemicals such as glass, Na2CO3 or NaOH as follows.
Here, the starting materials are H2SO4, NaCl, CaCO3 and C. NaOH and Cl2 are the main products. CaCO3 is converted to Ca(OH)2 as follows;
\(\beginIt is similar to the Leblanc process and the only difference is Mn 2+ (MnCl2) is recycled in Weldon but is wasted in Leblanc. Therefore, Weldon’s process is a cheaper method than the Leblanc process.
Similar to the above process, here also HCl is oxidised. But here the air is used to oxidise HCl in the presence of a catalyst (CaCl2).
\(\beginIn this process, NaOH and Cl2 are produced by electrolysis of Brine (NaCl) solution. The electrolytic cell contains both cathode and anode to be graphite rods and the electrolyte being NaCl solution. The construction is as follows.

At cathode: Na + + e – → Na
Other than the above reactions, some side reactions might also occur as follows.
\(\beginThe separation indicated in the diagram can be either asbestos or some plastic materials. The significance of the separation is to separate cathode and anode compartments and to avoid side reactions from occurring. A few modifications to the electrolytic cells yield the product more efficiently.
For example: Instead of carbon (graphite), if mercury is used as a cathode, efficiency is increased by up to 80 per cent, otherwise it will be arrow 20 per cent. Na + moving towards cathode mercury gets deposited as Na metal and reacts with Hg to form an amalgam, which can then be hydrolysed to form NaOH.
Na(amalgam) + H2O → NaOH + H2 + Hg.

With Acids
When sodium hydroxide is reacted with an acid (protic acids) it forms pure water and salts.
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
With Metals and Oxides
Sodium hydroxide can react with metals at high temperatures to form metal oxides. At temperatures above 500 °C, iron can react endothermically with sodium hydroxide to form iron(III) oxide, sodium metal, and hydrogen gas.
4 Fe + 6 NaOH → 2 Fe2O3 + 6 Na + 3 H2
However, some transition metals tend to react vigorously with NaOH. For example, aluminium can be used to precipitate transition metal hydroxides.
With Acidic Oxides
Sodium hydroxide can also react with acidic oxides. In such reactions, harmful acidic gases are purified. For example, the reaction with sulfur dioxide is given below.
NaOH is a chemical compound that is used in many instances. Some of its common uses are listed below.
When handling sodium hydroxide for use, especially bulk volumes, it should be stored carefully given the chemical’s burn hazard. Sodium hydroxide can be stored in bottles for small-scale laboratory use. For use in cargo handling and transport, it should be stored in intermediate bulk containers (medium volume containers). In manufacturing or wastewater plants, NaOH should be stored in large stationary storage tanks with volumes up to 100,000 gallons.
Some of the most common materials used in NaOH storage containers include carbon steel, polyvinyl chloride (PVC), stainless steel, polyethene, and fibreglass-reinforced plastic (FRP, with a resistant liner). Sodium hydroxide must be stored in airtight containers.
Test your Knowledge on Sodium hydroxide